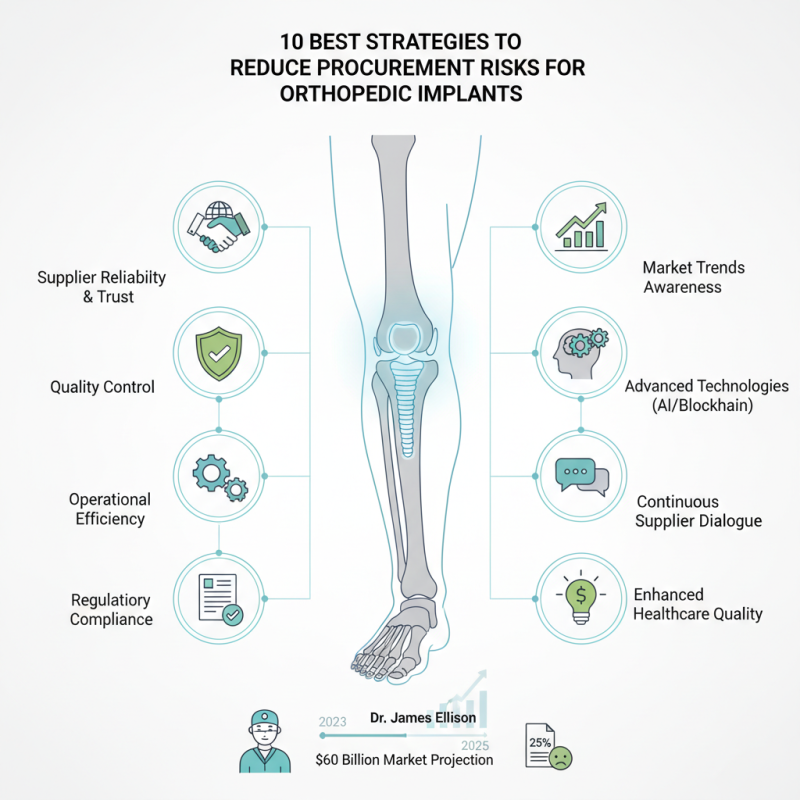
In the orthopedic implant industry, procurement risks have become a critical concern. With the market projected to reach $60 billion by 2025, organizations need effective strategies. Expert Dr. James Ellison notes, "Understanding the complexities of implant procurement is essential for success." This highlights the need for robust measures to minimize risks.
Challenges include supplier reliability and quality control. In a recent report, 25% of orthopedic surgeons expressed concerns over implant quality. Such issues can lead to serious patient outcomes and increased healthcare costs. Therefore, organizations must focus on how to reduce procurement risks for orthopedic implants to ensure patient safety and operational efficiency.
Companies must stay informed on the latest trends and regulations. Adopting advanced technologies can streamline procurement processes. Furthermore, continuous dialogue with suppliers is vital to build trust. Implementing these strategies can mitigate risks and enhance overall quality in orthopedic care.
Procurement risks in orthopedic implants pose significant challenges to healthcare providers. Recent studies reveal that nearly 20% of implant recalls occur due to quality control issues. This impacts patient safety and raises liability concerns. Understanding these risks is crucial for healthcare administrators.
Healthcare organizations must actively manage supply chain vulnerabilities. One key strategy is conducting thorough supplier audits. These audits can identify potential defects early. Regular assessments help ensure compliance with regulatory standards, reducing the chance of implant failures.
Tip: Establish a risk assessment team to analyze supplier data regularly.
The financial implications are substantial. Industry reports estimate that procurement risks can lead to losses exceeding $1 million annually for hospitals. This includes direct costs related to recalls and indirect costs such as patient care disruptions.
Tip: Invest in training staff on risk management practices related to procurement.
Engaging with multiple suppliers can also mitigate risks. Diversifying sources encourages competition, leading to better quality products. However, it complicates relationships and may require more oversight. Careful balance is needed to maintain effective supplier relationships.
Evaluating supplier reliability is crucial for reducing procurement risks in orthopedic implants. A data-driven approach helps in making informed decisions. Gathering historical data on suppliers can reveal patterns in delivery, quality, and compliance. This analysis provides insights into their performance over time, allowing for better risk management.
Tips for assessing supplier reliability include checking past performance records. Look for consistency in product quality over previous collaborations. Additionally, consider conducting surprise audits. This can unveil the true operational standards of a supplier. Regular communication is vital, too. Engaging with suppliers encourages transparency and accountability.
Data analytics tools can enhance reliability assessments. By using these tools, organizations can track relevant metrics efficiently. Ensure to review supplier financial health regularly. A supplier facing financial difficulties might struggle to meet commitments. Be prepared to switch suppliers if they do not meet expectations, no matter how established they might seem. Trust in the data; it often reveals what may not be apparent at first glance.
Implementing stringent quality control measures in supply chains is crucial for reducing procurement risks in orthopedic implants. These implants are critical medical devices. Ensuring they are safe and effective is non-negotiable. Quality control can involve thorough inspections and standardized testing protocols. Each component should meet the highest safety standards before reaching healthcare providers.
Moreover, regular audits of suppliers enhance transparency and trust. Engaging multiple suppliers can mitigate risks associated with single-source dependency. It creates competition among suppliers to maintain high standards. Additionally, traceability of materials is essential for accountability. If an issue arises, identifying the source of the problem becomes much easier.
However, challenges persist. Not all manufacturers may comply with stringent checks due to cost pressures. This reality can lead to subpar products reaching the market. Companies must reflect on their processes. Are they over-relying on assumptions about compliance? Adapting to new technologies for real-time monitoring can help. It’s vital to prioritize quality over speed in procurement to protect patients effectively.
In the ever-evolving field of orthopedic implants, leveraging technology is essential for risk mitigation. Advanced data analytics can enhance decision-making by providing real-time insights. Hospitals and procurement teams can analyze past performance, quality metrics, and supplier reliability instantly. Such information helps identify potential risks before they escalate. The use of artificial intelligence can also streamline vendor selection processes, ensuring only the most reliable suppliers are chosen.
Blockchain technology offers another layer of security. By providing a transparent record of transactions, it minimizes the risk of counterfeit implants. Every step, from manufacturing to delivery, can be tracked and verified. This level of transparency fosters trust among stakeholders. However, implementing such technologies requires careful planning and training. Not all teams may be ready for the transition, highlighting a gap that needs addressing.
Additionally, using digital collaboration tools can significantly improve communication between departments. Reducing misunderstandings and errors is crucial in procurement processes. Nonetheless, a reliance on technology should not overshadow the importance of human judgment. There will always be variables that technology cannot predict completely. Evaluating both technological solutions and human oversight is vital for a balanced approach to procurement risk management.
Building strong collaborative partnerships with key industry stakeholders is essential in reducing procurement risks for orthopedic implants. Such partnerships foster open communication and trust. When suppliers, manufacturers, and healthcare providers collaborate, they can share insights into evolving needs and market trends. This collaboration streamlines the procurement process, ensuring that each party is aligned with the latest standards and innovations.
Trust in relationships is pivotal. When stakeholders work together, they can address issues proactively. Regular meetings and feedback loops create a culture of transparency. However, not every partnership is smooth. Conflicting interests can arise, and communication may break down. It is crucial to evaluate these relationships regularly and make adjustments as needed. The goal is to cultivate a space where shared goals align with patient care and product quality.
In working towards better procurement practices, organizations must recognize the need for flexibility. As market conditions shift, so do the partnerships. Some alliances may need reevaluation. Not every collaboration will yield the desired results. A willingness to adapt and explore new partnerships can lead to better outcomes. Emphasizing flexibility ensures that procurement strategies remain robust and responsive to change.
| Strategy | Description | Potential Risks Mitigated | Stakeholder Involvement |
|---|---|---|---|
| Develop Robust Supplier Relationships | Foster long-term connections with suppliers to ensure reliability and quality. | Supply Chain Disruptions, Quality Assurance | Procurement Teams, Suppliers |
| Implement Strict Quality Control | Establish detailed quality assessments and audits of implant products. | Product Recalls, Compliance Issues | Quality Assurance Teams, Regulatory Agencies |
| Leverage Technology for Transparency | Use digital tools to enhance visibility throughout the supply chain. | Data Integrity, Supply Chain Visibility | IT Teams, Suppliers |
| Conduct Market Research | Regularly assess market trends and competitor strategies. | Market Fluctuations, Strategic Misalignment | Market Analysts, Procurement Teams |
| Ensure Compliance with Regulations | Stay updated on regulatory requirements in the medical device sector. | Legal Issues, Fines | Legal Teams, Compliance Officers |
| Diversify Supplier Base | Engage multiple suppliers to mitigate risks associated with single-source dependence. | Supply Chain Vulnerability, Price Increases | Procurement Teams, Strategy Teams |
| Negotiate Performance-Based Contracts | Create contracts that tie supplier payments to performance metrics. | Quality Issues, Delivery Delays | Procurement Teams, Suppliers |
| Engage in Collaborative Forecasting | Work with suppliers to predict demand more accurately. | Stockouts, Overproduction | Procurement Teams, Suppliers |
| Utilize Risk Assessment Tools | Employ tools to regularly assess and evaluate procurement risks. | Unforeseen Costs, Operational Disruptions | Risk Management Teams, Procurement Teams |
: Common risks include quality control issues, which can lead to recalls impacting patient safety.
Conduct thorough supplier audits to identify defects early and ensure compliance with standards.
They can lead to losses over $1 million annually from recalls and patient care disruptions.
Analyze historical data on suppliers to identify patterns in delivery, quality, and compliance.
Advanced analytics provide real-time insights, helping to identify risks before they escalate.
Regular communication and engagement with suppliers encourage transparency and accountability.
It encourages competition and improves quality, but may complicate relationships and require more oversight.
Surprise audits can reveal a supplier's true performance, ensuring operational standards are met.
Technology may not predict all variables; human judgment remains crucial in decision-making.
Establish a risk assessment team and invest in training staff on procurement practices.
To effectively answer the question of how to reduce procurement risks for orthopedic implants, it's essential to first understand the inherent risks associated with these medical devices. This involves evaluating supplier reliability through comprehensive, data-driven assessments that provide insights into their performance and stability. Implementing stringent quality control measures along the supply chain is another critical strategy, ensuring that every step meets established safety and efficacy standards.
Moreover, leveraging technology plays a significant role in enhancing risk mitigation in procurement processes. By utilizing advanced analytics and monitoring systems, organizations can proactively identify potential risks. Finally, establishing collaborative partnerships with key industry stakeholders fosters a supportive ecosystem for risk management, enabling better communication and shared best practices. These strategies collectively contribute to a robust procurement framework that minimizes risks in the orthopedic implant market.

 Nipa Orthopedic Implants
Nipa Orthopedic Implants