In the medical field, the safety and efficacy of orthopedic implants are paramount. This raises the question: why do orthopedic implants need strict batch management? Strict batch management entails rigorous tracking of production batches, materials used, and quality control processes. Such oversight ensures that each implant meets the highest safety standards before reaching patients.
Poor batch management can lead to complications. Instances of defective implants can result in surgeries being postponed or even serious health issues for patients. It is often overlooked how crucial it is to trace exact batch histories for recalls or investigations. This lack of awareness can foster skepticism regarding the reliability of orthopedic devices.
Moreover, healthcare professionals rely heavily on the assurance that implants are manufactured within strict guidelines. They depend on transparent processes to trust the devices they implant. The consequences of negligence in batch management can be severe. Thus, understanding the role of strict batch management becomes essential for the well-being of patients and the integrity of the orthopedic field.
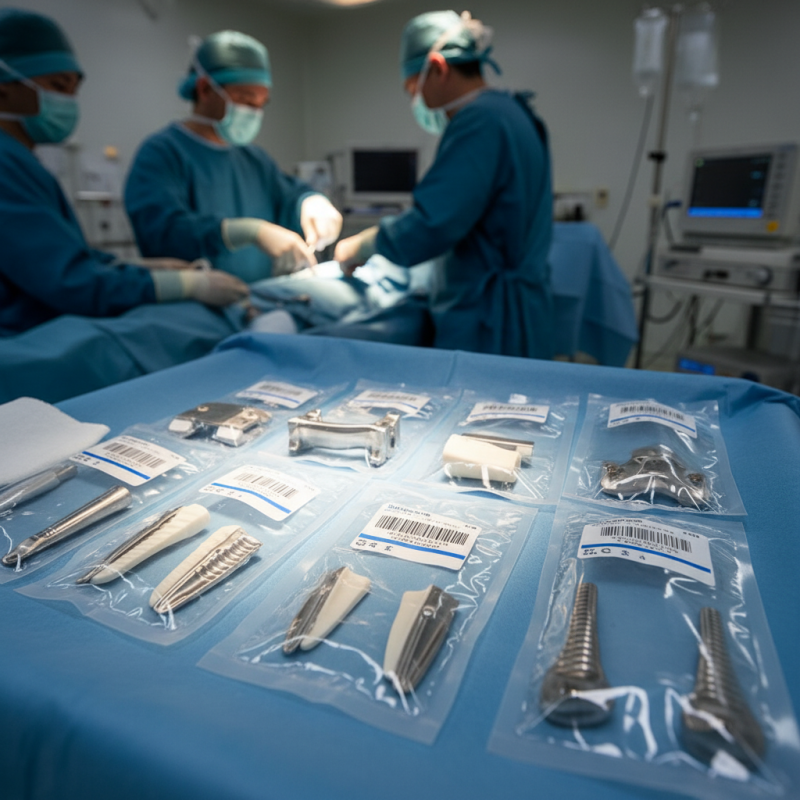
Strict batch management in orthopedic implants is critical for ensuring patient safety and product efficacy. With thousands of implants manufactured annually, the stakes are high. Proper tracking allows healthcare providers to trace each implant back to its production batch. This ensures that any potential defects are promptly identified and addressed. In cases of recalls, accurate records can save lives.
Implementing strict batch management also enhances quality control. Each batch undergoes rigorous testing for safety and effectiveness. If a problem arises, such as unexpected adverse reactions, it can be traced back to specific batches. This process not only safeguards patients but also bolsters the trust between manufacturers and medical professionals.
Tips: Always verify batch numbers before surgeries. Ensure staff are trained in tracking systems. Regular audits can help maintain accuracy. Remember, even small oversights can lead to significant consequences in patient care. Prioritizing batch management can prevent errors that compromise the entire surgical process.
Orthopedic implants play a crucial role in surgeries, making strict batch management vital. The regulation of these devices is more complex than many realize. Health authorities mandate tracking every production batch. This ensures safety and accountability throughout the supply chain.
Key regulations often focus on traceability. Manufacturers must maintain detailed records of each batch, including materials used and production dates. This practice helps identify issues promptly. If a defect arises, it limits the scope of recalls, protecting patients and hospitals alike. The challenge, however, lies in the consistent adherence to these guidelines, which can be cumbersome.
Quality assurance processes also support batch management practices. Regular audits are required, but implementing them can be difficult. Smaller firms may struggle due to resource constraints. Ensuring compliance becomes an ongoing effort rather than a one-time task. Reflecting on these challenges is essential for improving the overall management of orthopedic implants.
Effective batch management is crucial for orthopedic implants. Each implant batch must be tracked meticulously from production to implantation. This process ensures patient safety and enhances the longevity of the implants. Mismanagement can lead to serious complications. Even a minor oversight in batch tracking can result in harmful outcomes.
Patient safety hinges on the traceability of implants. In case of a recall, knowing the exact batch makes it easier to notify affected patients. This mitigates risks associated with defective implants. Additionally, well-managed batches contribute to the durability of orthopedic devices. Quality control during manufacturing is essential. It reduces the chance of implant failures, ensuring that patients remain active and pain-free.
However, maintaining strict batch management is challenging. It requires constant vigilance and dedicated resources. Many facilities struggle with this, often leading to lapses. These lapses can result in patient harm and increased healthcare costs. Implementing robust tracking systems is vital, yet not always feasible for every organization. Attention to detail must remain a priority in the surgical and management process.
Improper batch tracking in orthopedic implants can lead to devastating consequences. A statistical analysis revealed that the failure rates associated with poor batch management are alarmingly high. In one study, nearly 25% of reported failures linked to mislabeled or mismanaged batches resulted in serious complications for patients. This highlights the critical need for strict protocols in tracking and managing orthopedic implants.
Effective batch management ensures that healthcare providers use the correct implants. Every device must be accounted for, from manufacturing to patient usage. Problems arise when there is a lack of transparency in tracking these products. A 2021 report indicated that many practitioners could not identify implant batches during recalls due to inadequate records. Such situations put patient safety at risk and reflect systemic issues in the healthcare supply chain.
Regulations exist, but compliance remains inconsistent. Some manufacturers struggle with accurate tracking, while others have outdated systems. Healthcare professionals must advocate for better practices, but the process isn’t flawless. Despite advancements in technology, gaps in tracking systems often lead to confusion and mistakes. It is vital to recognize these shortcomings and work towards solutions that enhance accountability and safety in orthopedic care.
| Batch Number | Implant Type | Manufacture Date | Failure Rate (%) | Comments |
|---|---|---|---|---|
| B001 | Hip Implant | 2020-01-15 | 2.5 | Acceptable |
| B002 | Knee Implant | 2019-03-10 | 5.1 | High risk |
| B003 | Spinal Implant | 2021-06-22 | 1.8 | Optimal |
| B004 | Shoulder Implant | 2018-09-30 | 6.3 | Critical issue |
In orthopedic practices, strict batch management is crucial for ensuring patient safety and enhancing operational efficiency. Effective batch management involves meticulously tracking and documenting every implant used in procedures. This detail not only aids in compliance but also boosts traceability. When a recall occurs, having accurate batch records can minimize disruptions in patient care.
Implementing best practices requires training staff on the importance of batch management. Every team member should understand how to log implants properly. Regular audits can help identify gaps in tracking and documentation. Using digital tools can streamline the process, but reliance on technology shouldn't replace the need for human oversight. Mistakes can still happen, so fostering a culture of accountability is key.
Vigilance is necessary, as even small errors can lead to significant consequences. A misplaced implant record can cause confusion during surgeries. Additionally, staff may encounter challenges in adapting to new systems. Continuous feedback and adjustments are essential for developing a reliable batch management system that truly enhances patient safety and care quality.
: It ensures patient safety and product efficacy. Proper tracking helps identify and address defects promptly.
High failure rates can occur, leading to serious complications. Studies show nearly 25% of failures are linked to mismanagement.
Health authorities require detailed tracking of every production batch to ensure safety and accountability.
Always check batch numbers to avoid using incorrect implants. This simple step is vital for patient safety.
Consistent adherence to regulations is difficult. Smaller firms often struggle with resources for compliance.
Regular audits can improve tracking accuracy. However, some organizations find implementing them challenging.
Many cannot identify implant batches during recalls due to inadequate records, risking patient safety.
It creates transparency issues and complicates the identification of defects when problems arise.
Acknowledge the shortcomings in current systems. Work collaboratively to enhance accountability and safety measures.
Not entirely. Despite advances, gaps persist, leading to confusion in the management of orthopedic implants.
Strict batch management is vital for orthopedic implants due to its significant impact on patient safety and the longevity of the devices. Effective management practices ensure that each implant can be traced throughout its lifecycle, facilitating swift recalls if necessary. This traceability is governed by key regulations designed to maintain high standards in production and tracking. Without strict adherence to these protocols, failure rates can increase, leading to adverse patient outcomes and complications from poorly tracked implants.
Statistical analyses highlight that inadequate batch tracking is often linked to higher failure rates, emphasizing the necessity of implementing rigorous batch management systems. Best practices, such as comprehensive documentation and consistent monitoring, are essential for ensuring that orthopedic implants meet safety standards and perform optimally. Understanding "Why do orthopedic implants need strict batch management" underscores these practices as foundational components in safeguarding patient health and enhancing the reliability of orthopedic devices.

 Nipa Orthopedic Implants
Nipa Orthopedic Implants