Explore our flagship orthopedic implants designed for maximum osseointegration and stability.
 Cheap Price Titanium Alloy Coating Surgical Instrument Set Orthopedic Implant Cervical Interbody Fusion Peek Cage
Cheap Price Titanium Alloy Coating Surgical Instrument Set Orthopedic Implant Cervical Interbody Fusion Peek Cage
 Wholesale High Quality Titanium Coating Surgical Instrument Set Orthopedic Implant Cervical Interbody Fusion Peek Cage
Wholesale High Quality Titanium Coating Surgical Instrument Set Orthopedic Implant Cervical Interbody Fusion Peek Cage
 Titanium Coating Surgical Instrument Set Orthopedic Implant Cervical Interbody Fusion Peek Cage
Titanium Coating Surgical Instrument Set Orthopedic Implant Cervical Interbody Fusion Peek Cage
 Hindfoot Fusion Ankle Intramedullary Nail Titanium Interlocking Nail Exporters Hospital Surgical Equipment
Hindfoot Fusion Ankle Intramedullary Nail Titanium Interlocking Nail Exporters Hospital Surgical Equipment
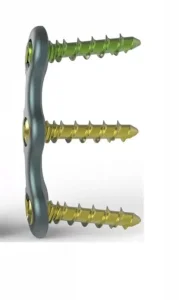
 Medical Equipment: Cervical Pedicle Screw System with Locking Plate
Medical Equipment: Cervical Pedicle Screw System with Locking Plate
 Medical Orthopedic Surgical Instruments Spine Posterior Cervical Screw Instrument Set
Medical Orthopedic Surgical Instruments Spine Posterior Cervical Screw Instrument Set
 Medical Orthopedic Surgical Instruments Spine Posterior Cervical Screw Instrument Set
Medical Orthopedic Surgical Instruments Spine Posterior Cervical Screw Instrument Set
 Anterior Cervical Fixation System with Screw for Cervical Spine with CE Certificate
Anterior Cervical Fixation System with Screw for Cervical Spine with CE Certificate
The global spinal fusion market is witnessing unprecedented growth, driven by an aging global population and the rising prevalence of degenerative disc diseases. Currently, North America and Europe lead in consumption, but the Asia-Pacific region is emerging as the fastest-growing hub for Cervical Fusion Cage manufacturing.
We are moving from traditional solid implants to "Biological active" structures. The industry is pivoting toward 3D-printed porous titanium and Titanium-coated PEEK (Polyetheretherketone) to combine the radiolucency of polymers with the osteoconductive properties of metals.
Safety is paramount. Top manufacturers are now adhering to stringent MDR (Medical Device Regulation) in Europe and FDA 510(k) clearances in the USA. Our factory implements a full-traceability system using AI monitoring to ensure every cage meets zero-defect standards.
The future of Cervical Fusion Cages lies in personalization. AI-driven imaging now allows factories to produce patient-specific cages that match the unique anatomy of a patient's vertebrae.
Additionally, surface modification technologies, such as plasma-sprayed titanium coating on PEEK, are becoming the "gold standard" for high-end orthopedic surgery, reducing the risk of cage subsidence and migration.

Hospitals and medical distributors worldwide face a critical need for stable, high-quality supply chains. From large-scale trauma centers in New York to specialized orthopedic clinics in Berlin, the demand for reliable cervical fusion solutions is universal.
Surgical departments prioritize "Set Completeness." Our factory provides not just the cage, but the entire surgical instrument set, ensuring seamless operation theater workflow.
Bulk buyers look for OEM/ODM flexibility. We offer customized laser marking and sterile packaging options that meet local health authority requirements across different continents.
Used in ACDF (Anterior Cervical Discectomy and Fusion) procedures, our cages provide immediate stability and a conducive environment for bone growth, catering to patients from athletes to the elderly.
Leading the world in medical grade precision manufacturing and cost-efficiency.
As a leading Cervical Fusion Cage manufacturer, we leverage China's advanced industrial ecosystem. This allows us to source medical-grade Titanium and PEEK materials at scale while maintaining ultra-precision CNC machining standards. Our AI-integrated quality control monitors every micron, ensuring that our "Made in China" orthopedic implants compete with the world's best at a fraction of the cost.

We have one of the most comprehensive orthopaedics portfolios in the world, we help heal and restore movement for millions of patients. Our products span various specialties, including joint reconstruction, trauma, craniomaxillofacial, spinal surgery, and sports medicine.
Building on our proud legacy of industry firsts, we are honoring our commitments to quality and innovation while creating a future where it's easier than ever before to keep people moving.
We are inspired to be good citizens of the world.
Our Credo challenges us to put the needs and well-being of the people we serve first. That means we are responsible to the communities in which we live and work and the global community as a whole.
We must be good citizens, encourage civic improvements, better health and education, and maintain the property we are privileged to use by protecting the environment and its natural resources.
At our core, supporting our partners in care is a key part of our mission. Whether that’s helping fund educational programs for the next generation of healthcare providers, providing medical kits for global healthcare missions, or supporting our industry partners to offer events for industry professionals, we’re dedicated to contributing to the advancement of healthcare across all verticals.
For more than 60 years, DePuy Synthes has partnered with the AO Foundation aiming to improve patient outcomes and build on our commitment to product and surgical innovation.
Advanced spine fixation systems and specialized instrument sets.
 Anterior Cervical Instrument Set Orthopedic Medical Set Surgical Instrument Medical Equipment
Anterior Cervical Instrument Set Orthopedic Medical Set Surgical Instrument Medical Equipment
 Spine Fixation Anterior Cervical Plate System
Spine Fixation Anterior Cervical Plate System
 China Manufacture Orthopedic Surgical Implants Laminar Hook for Posterior Cervical Fixation System Spinal Implant
China Manufacture Orthopedic Surgical Implants Laminar Hook for Posterior Cervical Fixation System Spinal Implant
 China Manufacture Orthopedic Surgical Implants Laminar Hook for Posterior Cervical Fixation System Spinal Implant
China Manufacture Orthopedic Surgical Implants Laminar Hook for Posterior Cervical Fixation System Spinal Implant
 Titanium Alloy Anatomical Hook Guide Pin for Anterior Cervical Plate Fixation Surgery
Titanium Alloy Anatomical Hook Guide Pin for Anterior Cervical Plate Fixation Surgery
 Pedicle Screw Fixation System Orthopaedic Spinal Implants Posterior Cervical Surgery Orthopedic Spine Surgical Instruments Set
Pedicle Screw Fixation System Orthopaedic Spinal Implants Posterior Cervical Surgery Orthopedic Spine Surgical Instruments Set
 Master 10 Spinal System,Titanium Alloy, Orthopedic Implant, Spine, Surgical, Medical Instrument Set, with CE/ISO/FDA, Dislocation, Fracture, Lumber and Cervical
Master 10 Spinal System,Titanium Alloy, Orthopedic Implant, Spine, Surgical, Medical Instrument Set, with CE/ISO/FDA, Dislocation, Fracture, Lumber and Cervical
 Disposable Medical Surgical Instrument Orthopedic Implant Titanium Coating Banana Lumbar Peek Interbody Fusion Cage Spine System
Disposable Medical Surgical Instrument Orthopedic Implant Titanium Coating Banana Lumbar Peek Interbody Fusion Cage Spine System
Choosing a Cervical Fusion Cage manufacturer is a decision that impacts surgical success and patient recovery times. In the modern medical landscape, the choice of material—whether PEEK, Titanium, or a hybrid—defines the performance of the implant. PEEK cages have long been favored for their modulus of elasticity, which is similar to human bone, thereby reducing the "stress shielding" effect. However, the inert nature of PEEK sometimes leads to a lack of integration at the bone-implant interface.
This is where our factory’s innovation comes in. By applying a porous titanium coating via physical vapor deposition (PVD) or plasma spray, we provide the best of both worlds: the radiolucent properties of PEEK for clear post-operative imaging and the bioactive surface of titanium for rapid osseointegration. As a Top 10 factory, our production line is equipped with high-precision Swiss-type CNC machines and robotic polishing systems to ensure that every serration and thread is anatomically perfect.
Looking at the global industrial status, the shift towards minimally invasive surgery (MIS) is dictating the design of the next generation of cages. Expandable cervical cages and those with integrated fixation screws (Stand-alone cages) are becoming increasingly popular. These designs reduce the need for additional plating, thereby shortening operative time and reducing tissue trauma. Our R&D team works closely with surgeons in Europe and Southeast Asia to refine these designs for diverse localized application scenarios.
For international procurement officers, sourcing from a Chinese factory offers more than just price advantages. It offers agility. While traditional Western giants have long lead times, our factory utilizes AI-driven supply chain management to reduce production cycles by 40%. We maintain a massive inventory of standard sizes (6mm to 12mm heights) ready for immediate global shipping, ensuring that your hospital never faces a shortage during critical surgical windows.
Furthermore, we understand the importance of educational support. Following the legacy of industry leaders, we provide comprehensive surgical guides and 3D-animated training videos for every instrument set we manufacture. We believe that a high-quality product is only as good as the surgeon's ability to use it effectively. By choosing us, you are not just buying a medical device; you are partnering with a manufacturer dedicated to advancing the future of spinal care through technology, empathy, and industrial excellence.