Global Industrial Leadership in Craniomaxillofacial Fixation & Orthopedic Innovation
 Orthopedic Surgery Trauma Bone Pin Stent Spanning External Fixator for Tibia Femur
Orthopedic Surgery Trauma Bone Pin Stent Spanning External Fixator for Tibia Femur
 Headless Compression Hollow Screw Cannulated Screw Bone Screws Orthopedic Implants Bone Implant
Headless Compression Hollow Screw Cannulated Screw Bone Screws Orthopedic Implants Bone Implant
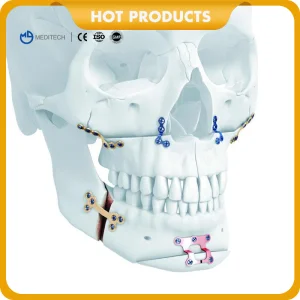 Titanium Cmf Locking Plates and Screws-Maxillofacial Orthopedic Implant System
Titanium Cmf Locking Plates and Screws-Maxillofacial Orthopedic Implant System
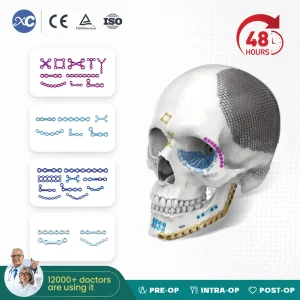 Cmf System Titanium Material Orthopedic Surgery Maxillofacial Locking Plates and Screws Implant
Cmf System Titanium Material Orthopedic Surgery Maxillofacial Locking Plates and Screws Implant
 Orthopedic External Fixator for Bone Fractures
Orthopedic External Fixator for Bone Fractures
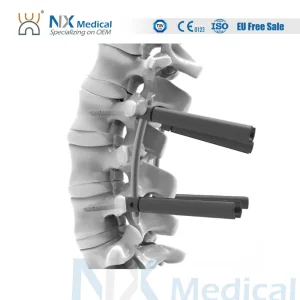 Nx Medical Cannulated Bone Cement Spinal Pedicle Screw 5.5mm System Bone Fixation
Nx Medical Cannulated Bone Cement Spinal Pedicle Screw 5.5mm System Bone Fixation
 Nx Medical Sf-IV Mis Cannulated Bone Cement Spinal Pedicle Screw 5.5 for Bone Fixation
Nx Medical Sf-IV Mis Cannulated Bone Cement Spinal Pedicle Screw 5.5 for Bone Fixation
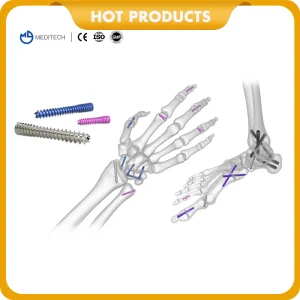 Orthopedic Implants Titanium Medical Bone Screws Cannulated Headless Compression Screw
Orthopedic Implants Titanium Medical Bone Screws Cannulated Headless Compression Screw
The Occipital Bone Plate market has witnessed an unprecedented evolution in the last decade. As neurosurgery and spinal orthopedic surgery become increasingly refined, the demand for stabilization systems that bridge the transition between the skull and the cervical spine has surged. These specialized plates are critical in managing craniocervical instability, trauma, and reconstructive procedures following tumor resections.
Today, the Occipital Bone Plate industry is no longer confined to basic mechanical fixation. The commercial landscape is shifting toward personalized medicine. Large-scale manufacturers are integrating CAD/CAM technologies to produce patient-specific implants. The global market is dominated by North America and Europe, but the Asia-Pacific region, led by China, is rapidly becoming the manufacturing hub for the world's orthopedic supply chain due to advancements in medical-grade titanium processing.
The next frontier in Occipital Bone Plate manufacturing involves AI-driven design. Algorithms now analyze pre-operative CT scans to suggest the optimal plate thickness and screw trajectory, reducing surgery time by up to 30%. Furthermore, the industry is exploring PEEK (Polyetheretherketone) and carbon-fiber composites to provide radiolucency, allowing surgeons to monitor bone healing more effectively without metal artifact interference.
A powerhouse in the J&J MedTech portfolio. Known for their Synapse System, they provide unparalleled versatility in posterior cervical and occipital fixation.
The Vertex Select Reconstructive System is a benchmark in the industry, offering a multi-axis screw design that simplifies craniocervical junction stabilization.
Stryker’s craniomaxillofacial division focuses on low-profile plate designs that minimize soft tissue irritation while maintaining extreme rigid fixation.
Renowned for the Ellipse system, Globus specializes in streamlined instrumentation that caters to minimally invasive surgical approaches.
Offering comprehensive solutions for neurosurgical trauma, Zimmer Biomet is a leader in high-durability titanium alloy plates with specialized coating technologies.
The Aesculap division brings German engineering to the occipital region, focusing on biomechanical stability and long-term implant integration.
Highly focused on the "Integrated Spine Platform," their occipital solutions are designed for seamless transition from the skull to the lower cervical spine.
A leader in bone growth stimulation and fixation, providing unique solutions for complex occipital-cervical fusion cases.
A rising titan in the global market, MicroPort represents the pinnacle of high-tech manufacturing in China, competing directly with Western giants.
Specializing in OEM/ODM solutions, we offer high-precision Occipital plates that meet international CE/ISO standards with a focus on cost-efficiency.
Hospitals and medical distributors today are not just looking for a product; they are looking for a sustainable supply chain. In regions like Southeast Asia and Latin America, the focus is on "Quality-to-Cost" ratios. This is where localized application scenarios become vital.
In trauma centers, Occipital Bone Plates must be ready for "off-the-shelf" use. In elective reconstructive surgery, surgeons prefer customized plates that mirror the patient's unique anatomy. Our factory supports both requirements, providing modular kits and bespoke designs.
Modern procurement departments prioritize factories that offer transparent certification (FDA/CE/ISO), rapid lead times (under 2 weeks), and ecological packaging. The shift is moving away from sole-source procurement to a more diversified, resilient supply model.
Our facility utilizes 5-axis CNC machining centers from Switzerland and Japan to ensure every Occipital Bone Plate meets tolerances within microns. We use only Grade 5 ELI Medical Titanium.
From initial concept to a 3D-printed prototype, our R&D cycle is 50% faster than the industry average. This allows our partners to respond to market shifts in real-time.
Every single plate undergoes a 100% inspection process, including fatigue testing, surface finish analysis, and chemical composition verification. Our zero-defect policy is our pride.
Located in China's industrial heartland, we have direct access to raw material sources and international logistics hubs, ensuring that global shipping remains unaffected by regional disruptions.
We have one of the most comprehensive orthopaedics portfolios in the world, we help heal and restore movement for millions of patients. Our products span various specialties, including joint reconstruction, trauma, craniomaxillofacial, spinal surgery, and sports medicine.
Building on our proud legacy of industry firsts, we are honoring our commitments to quality and innovation while creating a future where it's easier than ever before to keep people moving.
We are inspired to be good citizens of the world.
Our Credo challenges us to put the needs and well-being of the people we serve first. That means we are responsible to the communities in which we live and work and the global community as a whole. We must be good citizens, encourage civic improvements, better health and education, and maintain the property we are privileged to use by protecting the environment and its natural resources.


DePuy Synthes and the AO Foundation Partnership: For more than 60 years, DePuy Synthes has partnered with the AO Foundation aiming to improve patient outcomes and build on our commitment to product and surgical innovation.
At DePuy Synthes, supporting our partners in care is a key part of our mission. Whether that’s helping fund educational programs for the next generation of healthcare providers, providing medical kits for global healthcare missions, or supporting our industry partners to offer events for industry professionals, we’re dedicated to contributing to the advancement of healthcare across all verticals.
 Orthopedic Implants Titanium Medical Bone Screws Cannulated Headless Compression Screw
Orthopedic Implants Titanium Medical Bone Screws Cannulated Headless Compression Screw
 Orthopedic Implants Maxillofacial Locking Reconstruction H Bone Plate Surgical for Bone Surgery
Orthopedic Implants Maxillofacial Locking Reconstruction H Bone Plate Surgical for Bone Surgery
 Orthopedic Implants Maxillofacial Locking Reconstruction H Bone Plate Surgical for Bone Surgery
Orthopedic Implants Maxillofacial Locking Reconstruction H Bone Plate Surgical for Bone Surgery
 Orthopedic Bone Surgical Set 6.0mm Spinal Pedicle Screw System Instrument Set
Orthopedic Bone Surgical Set 6.0mm Spinal Pedicle Screw System Instrument Set
 Factory Direct Supply Orthopedic Spine Implant Bone Cement-Injectable Cannulated Pedicle Screws High Quality
Factory Direct Supply Orthopedic Spine Implant Bone Cement-Injectable Cannulated Pedicle Screws High Quality
 Wrist External Fixator Hand Bone Pin Stent Radius External Fixator
Wrist External Fixator Hand Bone Pin Stent Radius External Fixator
 Orthopedic Plates Titanium Medical Surgical Fracture Bone Trauma Large Fragment Proximal Femur Neck Plate with Hook Locking Plate with CE
Orthopedic Plates Titanium Medical Surgical Fracture Bone Trauma Large Fragment Proximal Femur Neck Plate with Hook Locking Plate with CE
 Hot Selling Human Orthopedic Surgical Plates Ulna Olecranon Locking Plates/Implants
Hot Selling Human Orthopedic Surgical Plates Ulna Olecranon Locking Plates/Implants