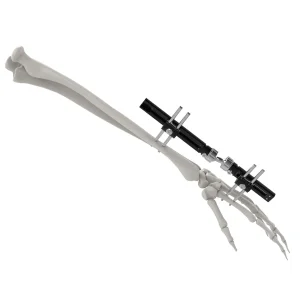
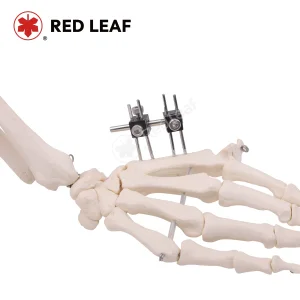
Engineered for Precision in Finger and Phalange Reconstructions



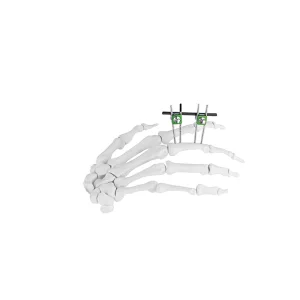
The orthopedic device landscape in the United States is currently undergoing a massive technological shift. As a leading hub for medical innovation, US-based surgeons are increasingly demanding Mini External Fixators that offer both high stability and minimally invasive application. In states like California, Massachusetts, and Florida—recognized global centers for biotech—the application of small bone fixators has moved beyond simple trauma to include complex diabetic foot salvage and pediatric limb lengthening.
Current trends show a 15% annual increase in the use of unilateral and circular mini-frames for phalange and metacarpal fractures across Level 1 Trauma Centers in the US. This surge is driven by the rise in outpatient surgical centers (ASCs) where Mini Fixator Systems allow for quicker recovery times and reduced hospital stays.
Our commitment to quality aligns with the rigorous standards of the American orthopedic market, ensuring that every screw, rail, and clamp meets the durability required for active US patient demographics.

Tailored Solutions for Local Healthcare Challenges
From NFL hand injuries to high-impact collegiate athletics, our Mini Rail Systems provide the rigid stabilization needed for metacarpal fractures in athletes, promoting early mobilization.
With millions of Americans affected by diabetes, our Foot External Fixators are critical in treating Charcot arthropathy and stabilizing small bone structures during ulcer healing.
Our ultra-lightweight titanium mini-fixators are designed for the delicate bones of pediatric patients, ensuring growth plate protection during fracture reduction.

We have one of the most comprehensive orthopaedics portfolios in the world, we help heal and restore movement for millions of patients. Our products span various specialties, including joint reconstruction, trauma, craniomaxillofacial, spinal surgery, and sports medicine.
We are inspired to be good citizens of the world. Building on our proud legacy of industry firsts, we are honoring our commitments to quality and innovation while creating a future where it's easier than ever before to keep people moving.
Our Credo challenges us to put the needs and well-being of the people we serve first. That means we are responsible to the communities in which we live and work and the global community as a whole.
The United States orthopedic fixation market is projected to reach several billion dollars by 2030, with Mini External Fixation systems occupying a significant niche. As a factory and supplier, we understand that US surgeons prioritize radiolucent materials and modular designs. Our systems utilize high-grade Aluminum and Stainless Steel alloys that are compatible with advanced imaging systems found in top US medical institutions like the Mayo Clinic or Johns Hopkins.
Furthermore, the regulatory environment in the US, governed by the FDA, demands transparency in manufacturing. Our production lines are optimized to provide full traceability, from raw material sourcing to the final sterile packaging. This makes us a preferred supplier for large-scale orthopedic distributors across the East and West Coasts.
Our "Mini-Fixator" series for Hand and Foot surgery is designed with the American lifestyle in mind—durable enough for heavy-duty vocational rehabilitation and precise enough for the finest cosmetic outcomes in phalangeal realignment.
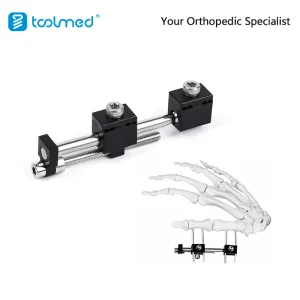
Explore our full range of small bone implants and instruments

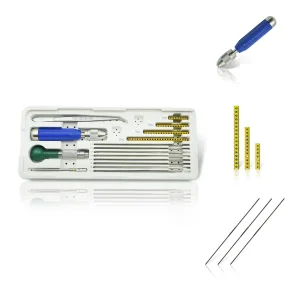




As we look toward 2025 and beyond, the integration of AI and Robotics into orthopedic surgery is no longer a futuristic dream but a reality in the United States. Our R&D team is currently exploring "Smart Fixators" equipped with micro-sensors that can monitor bone healing progress in real-time, transmitting data to a surgeon’s smartphone through secure cloud networks. This innovation is particularly relevant for the US market, where telehealth and remote patient monitoring are seeing unprecedented growth.
Material science is also evolving. We are shifting from traditional stainless steel to ultra-lightweight carbon fiber and 3D-printed titanium structures. These materials offer a "modulus of elasticity" closer to that of human bone, reducing the risk of stress shielding—a common complication in traditional fixation. For hand surgeons in Los Angeles or New York, this means their patients can return to work and daily activities faster, with less bulky hardware attached to their extremities.
Moreover, the environmental impact of medical manufacturing is a growing concern for US healthcare providers. Our factory has implemented "Green Manufacturing" protocols, reducing waste in the production of Mini External Fixator components by 30% over the last two years. We believe that being a "good citizen of the world" means protecting our environment while saving lives.
For US-based surgical teams, we provide comprehensive training modules, including VR (Virtual Reality) surgical simulations, allowing residents and veteran surgeons to master the application of our Combined Type Fixators and Rail Systems before entering the OR. This commitment to education is what differentiates us as a premium supplier in the competitive American market.
Ready to upgrade your trauma inventory with FDA-compliant solutions?
Send Inquiry Now & Get Expert Consultation