




1 / 5










| Still deciding? Get samples of $ ! US$ 48/Piece Request Sample |
| Product Number | Product Name | Material | Size (mm) |
|---|---|---|---|
| 73601 | KCO Laminar Hook 736 | Titanium Alloy | 4.5mm |
| 73602 | 6mm |

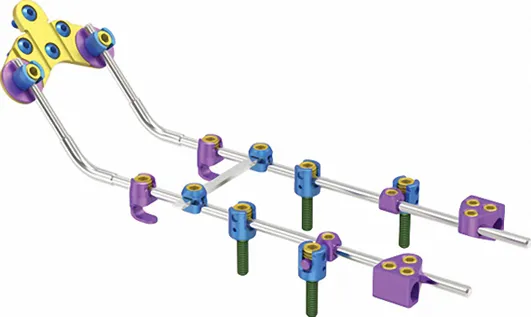

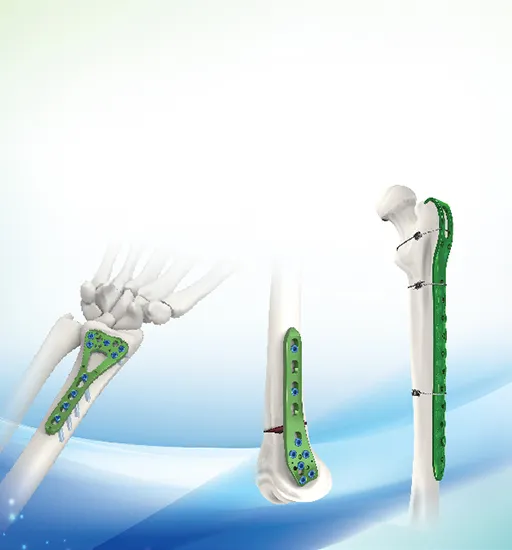


As a leading manufacturer of orthopedic implants and instruments established in 1958, the focus remains on high-quality production including trauma plates, interlocking nails, spinal fixators, external fixators, and power tools. The facility operates under strict quality control standards to serve global markets.

 Nipa Orthopedic Implants
Nipa Orthopedic Implants