



1 / 5










| Still deciding? Get samples of $ ! US$ 13.6/Piece Request Sample |



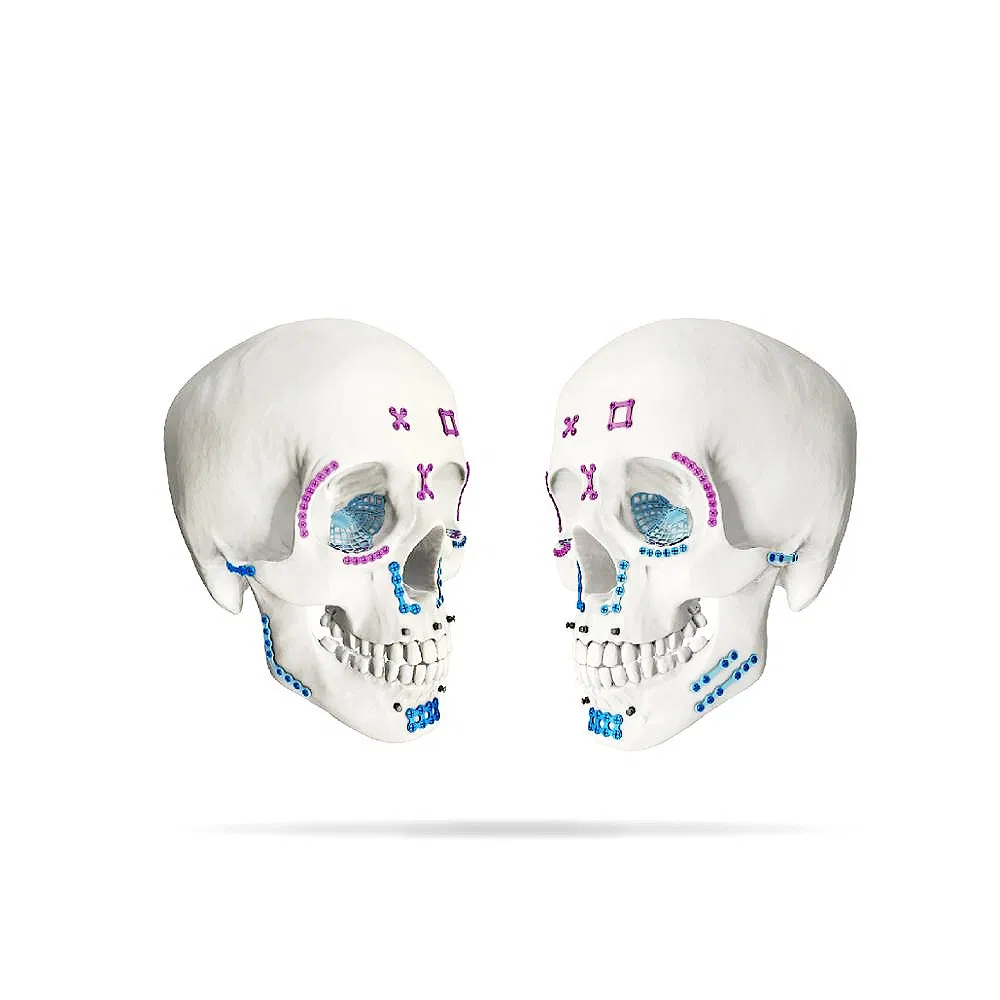
Screw Size Compatibility: 2.0mm Self-Tapping & 2.0mm Self-Drilling Screws.
Indications: Nasal part, Pars orbitalis, Pars zygomatica, Maxilla region, Mandible (simple and stable trauma).
| Order Code | Holes | Specification |
|---|---|---|
| 10.01.03.04115020 | 4 holes, 1.0mm, Left | Length 19 mm |
| 10.01.03.04215020 | 4 holes, 1.0mm, Right | Length 19 mm |
| 10.01.03.04115024 | 4 holes, 1.0mm, Left | Length 23 mm |
| 10.01.03.04215024 | 4 holes, 1.0mm, Right | Length 23 mm |


Adequate Inventory - Shipping Within 1-5 Days





 Nipa Orthopedic Implants
Nipa Orthopedic Implants