

1 / 3






| Customization: | Available |
|---|---|
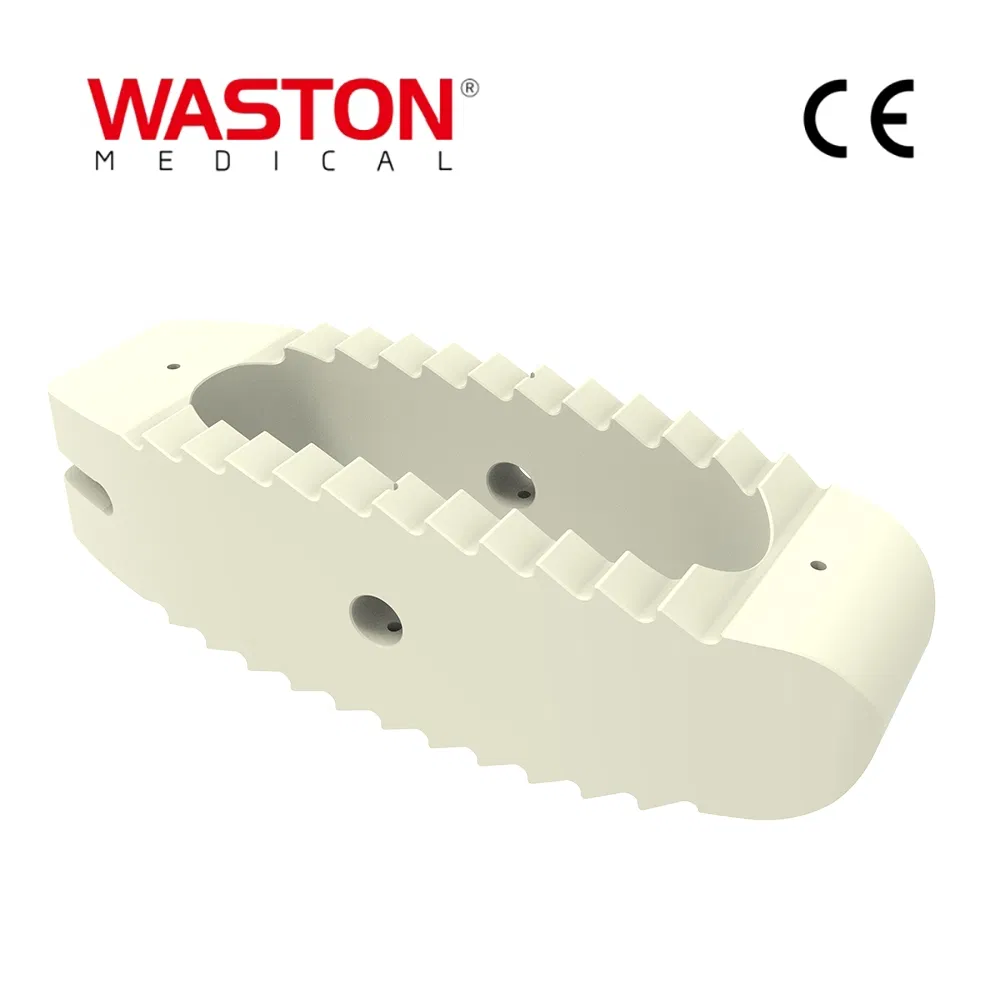
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |

| PRODUCT NAME | PRODUCT CODE | DIA | DATA |
|---|---|---|---|
| Laminoplasty Plate | 11101012 | / | H=12 |
| 11101014 | / | H=14 | |
| 11101016 | / | H=16 | |
| Screw (Cross self-tapping) | 10511306 | φ2.5 | L=6 |
| 10511308 | φ2.5 | L=8 | |
| 10511310 | φ2.5 | L=10 |


As a national high-tech enterprise, R&D investment exceeds 8% of total sales annually. We specialize in transforming clinical experience into innovative medical solutions with numerous national patents.
Equipped with German and American machining centers and Japanese automatic lathes, our quality control system meets ISO13485 and CE MDD 93/42/EEC standards.




 Nipa Orthopedic Implants
Nipa Orthopedic Implants