
1 / 5










| Customization: | Available |
|---|---|
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
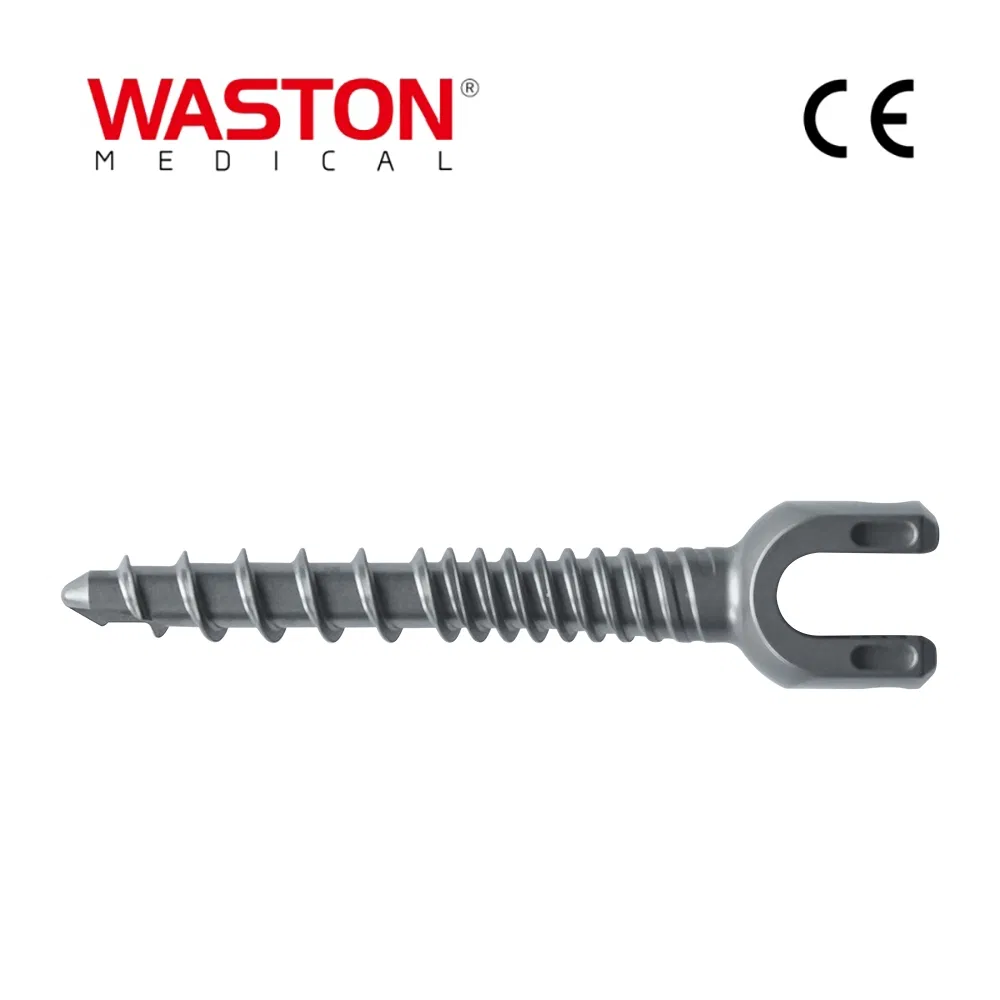
| PRODUCT NAME | PRODUCT CODE | DIA | DATA |
|---|---|---|---|
| Laminoplasty Plate | 11101012 | / | H=12 |
| 11101014 | / | H=14 | |
| 11101016 | / | H=16 | |
| Screw (Cross self-tapping) | 10511306 | φ2.5 | L=6 |
| 10511308 | L=8 | ||
| 10511310 | L=10 | ||
| Screw (Self-tapping & drilling) | 10511406 | φ2.5 | L=6 |
| 10511408 | L=8 | ||
| 10511410 | L=10 |

The manufacturing process operates strictly according to the ISO13485 quality system, adhering to medical devices manufacturing quality management, implants, and sterile medical devices execution rules, as well as the CE MDD 93/42/EEC directive.



Investment in state-of-the-art equipment includes German-imported and American-imported machining centers and Japanese-imported automatic lathes. The testing center features a biomechanical laboratory and a physical-chemical testing center for comprehensive product analysis.


 Nipa Orthopedic Implants
Nipa Orthopedic Implants