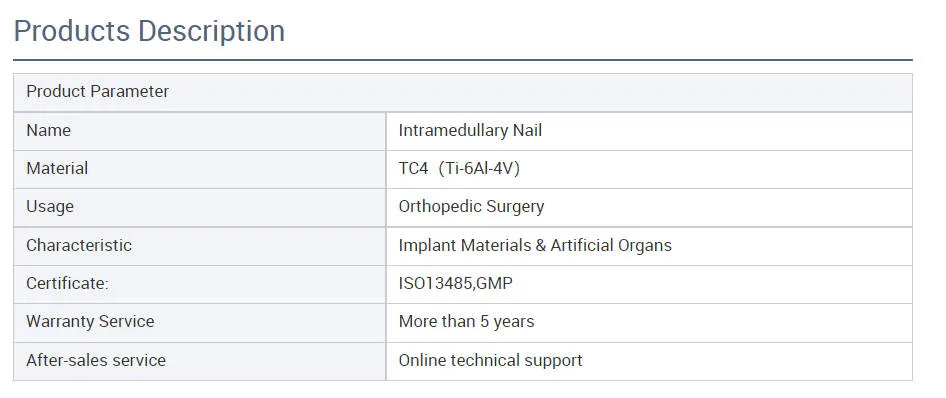
Product Description
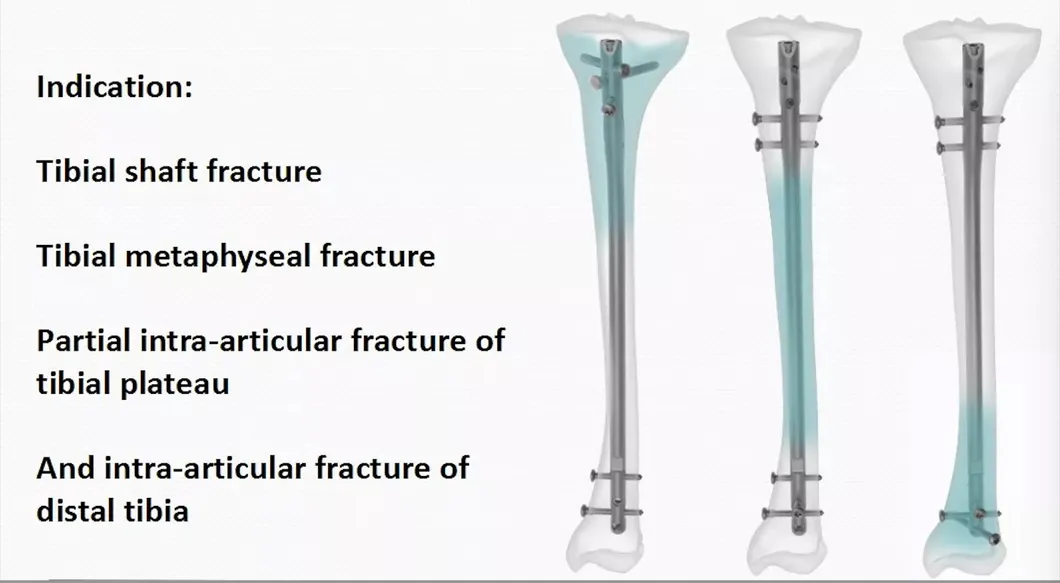
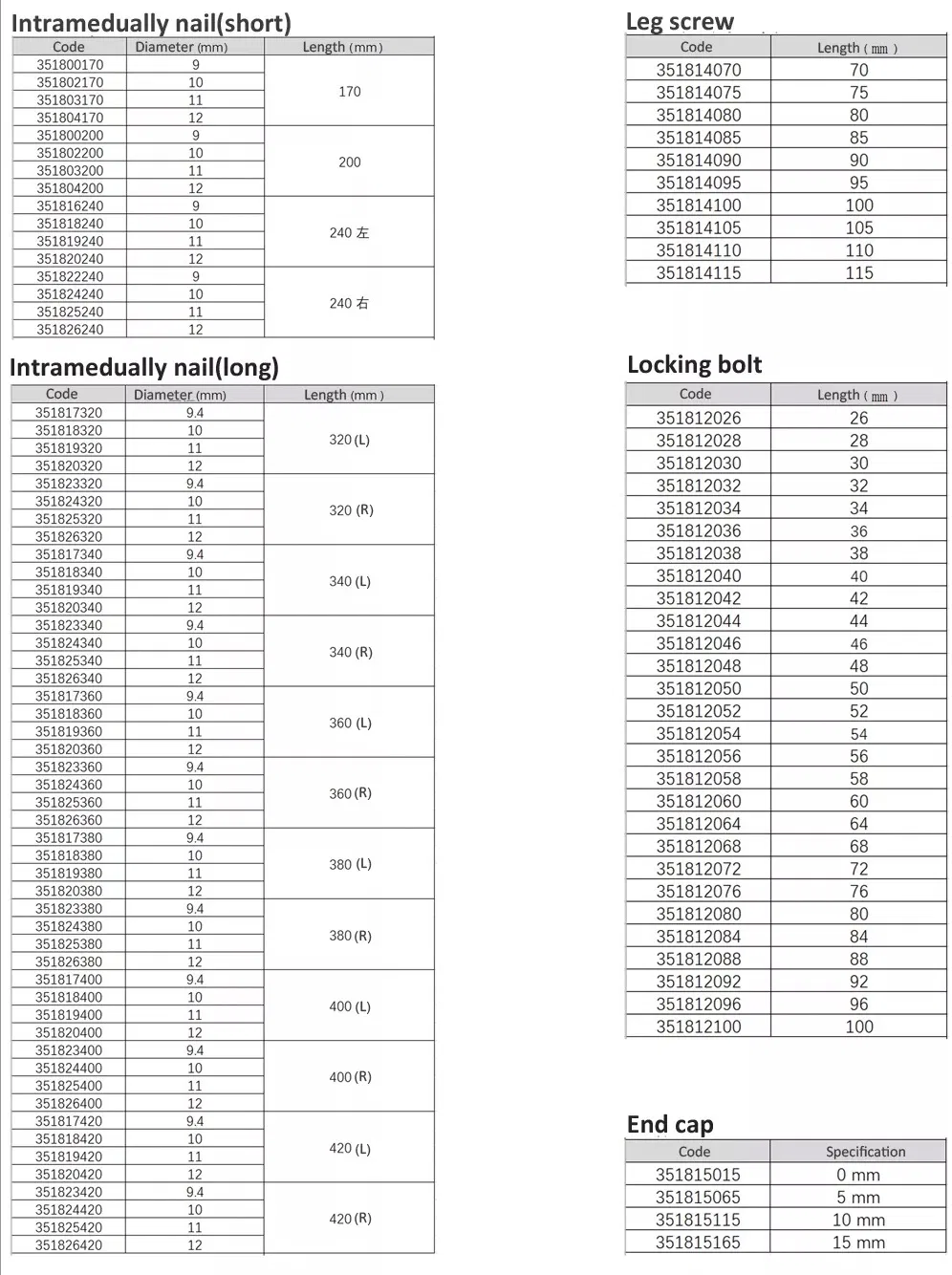
Tibia Intramedullary Nail System
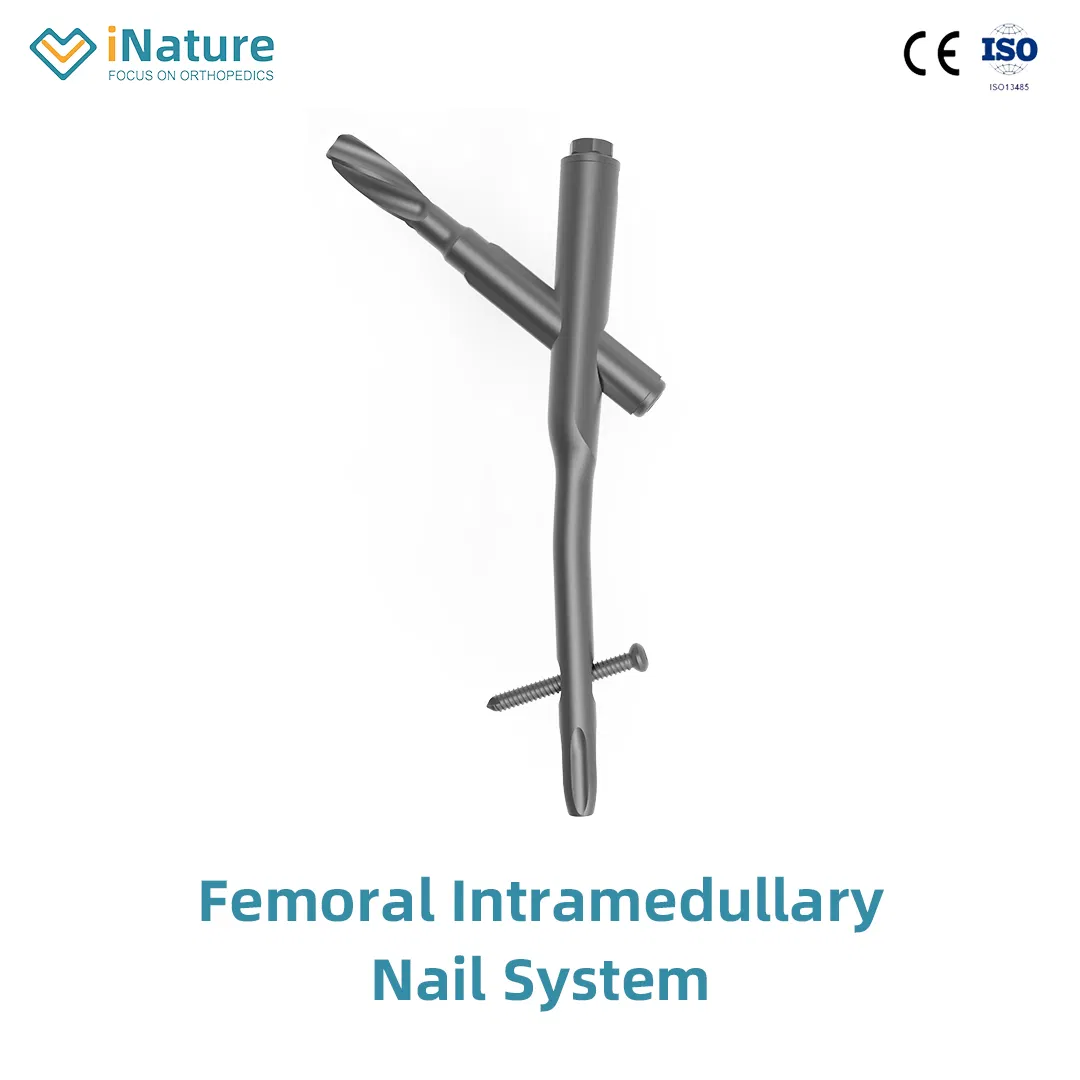
Femur Intramedullary Nail System
✔
Premium Material: High-grade Titanium with Oxidation/Milling surface finish.
✔
Anatomical Design: 5° medial-lateral angle allows insertion at the tip of the greater trochanter.
✔
Stress Reduction: Lateral flatten design reduces stress to the lateral cortical bone.
✔
Elastic Groove Tip: Eases insertion and minimizes concentration stress.
✔
Curved Design: 240mm distal curved design prevents nail tip contact with cortical bone, reducing pain.
✔
Advanced Anchoring: Lag screw insertion provides excellent compaction, ideal for osteoporotic patients.
✔
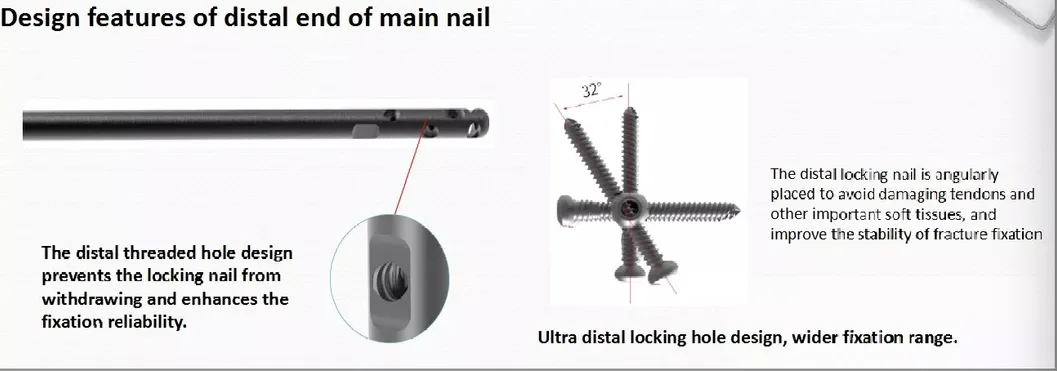
Efficiency: Locking bolt with double thread design significantly reduces operation time.
Frequently Asked Questions
What is the primary material used in these implants?
The intramedullary nail systems are primarily manufactured from high-quality Pure Titanium, ensuring excellent biocompatibility and strength.
Do the products carry international quality certifications?
Yes, our orthopedic implants are fully compliant with CE and ISO 13485 standards, meeting Class III medical instrument classifications.
What kind of after-sale support and warranty is provided?
We offer a one-year warranty and ongoing online technical support. We guarantee exchange or repair for quality issues identified within the first month and year respectively.
Is the packaging sterile for immediate surgical use?
Yes, all products are provided in a Sterile Package to ensure safety and convenience for medical procedures.
Can you handle custom OEM orders for large quantities?
Absolutely. We accept OEM orders and have a robust production capacity of 2000 pieces per month to meet large-scale global demands.
How does the design benefit osteoporotic patients?
The lag screw insertion provides superior compaction to cancellous bone, and the wide surface area of the tip offers better anchoring specifically for patients with low bone density.




















 Nipa Orthopedic Implants
Nipa Orthopedic Implants