














| Customization: | Available |
|---|---|
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
Semi-Ring Combined Fixator
1 Indication: Primarily used for fractures around the hip joint.
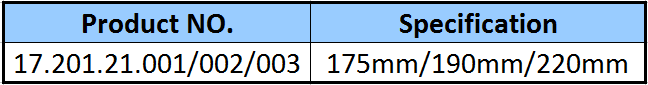
2 Design: Specially designed for tibial plateau fractures and distal tibial fractures. It is employed in conjunction with 3 to 4 olive-head Kirschner wires and a 4.8mm drill bit.
3 Screw Compatibility: Available in 6.0/5.0 screws (SS/TA).
As a national high-tech enterprise, R&D investment has consistently exceeded 8% of total sales, enabling the conversion of clinical expertise and innovative ideas into practical achievements. We've launched several groundbreaking products with national invention and utility model patents.
We lead with revolutionary technology including: the Rev Drill system, the Three-Row Stapler, Sternal Fixation and Rib Plate. These products exemplify leadership and innovation benchmarks in the medical device industry.

Our core philosophy is 'Good faith as the base, quality above all.'
We adhere rigorously to the ISO13485 quality system, conforming to medical device manufacturing quality management specifications, implant and sterile medical device execution rules, and the CE MDD 93/42/EEC directive.
Advanced equipment includes:
Since 2011, over 20 million RMB has been invested in our Testing Center, featuring a biomechanics laboratory and a physical-chemical testing center for raw material procurement control, product quality control, and new product performance analysis.

Our products are exported to numerous countries, including Colombia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. Feedback confirms that our product quality meets or exceeds national standards.









We are a major OEM supplier for some of the largest global players in the medical device industry.
 Nipa Orthopedic Implants
Nipa Orthopedic Implants