




1 / 5










| Still deciding? Get samples of $ ! US$ 300/Set Request Sample |

| Product Component | Model | Specification Details | Material |
|---|---|---|---|
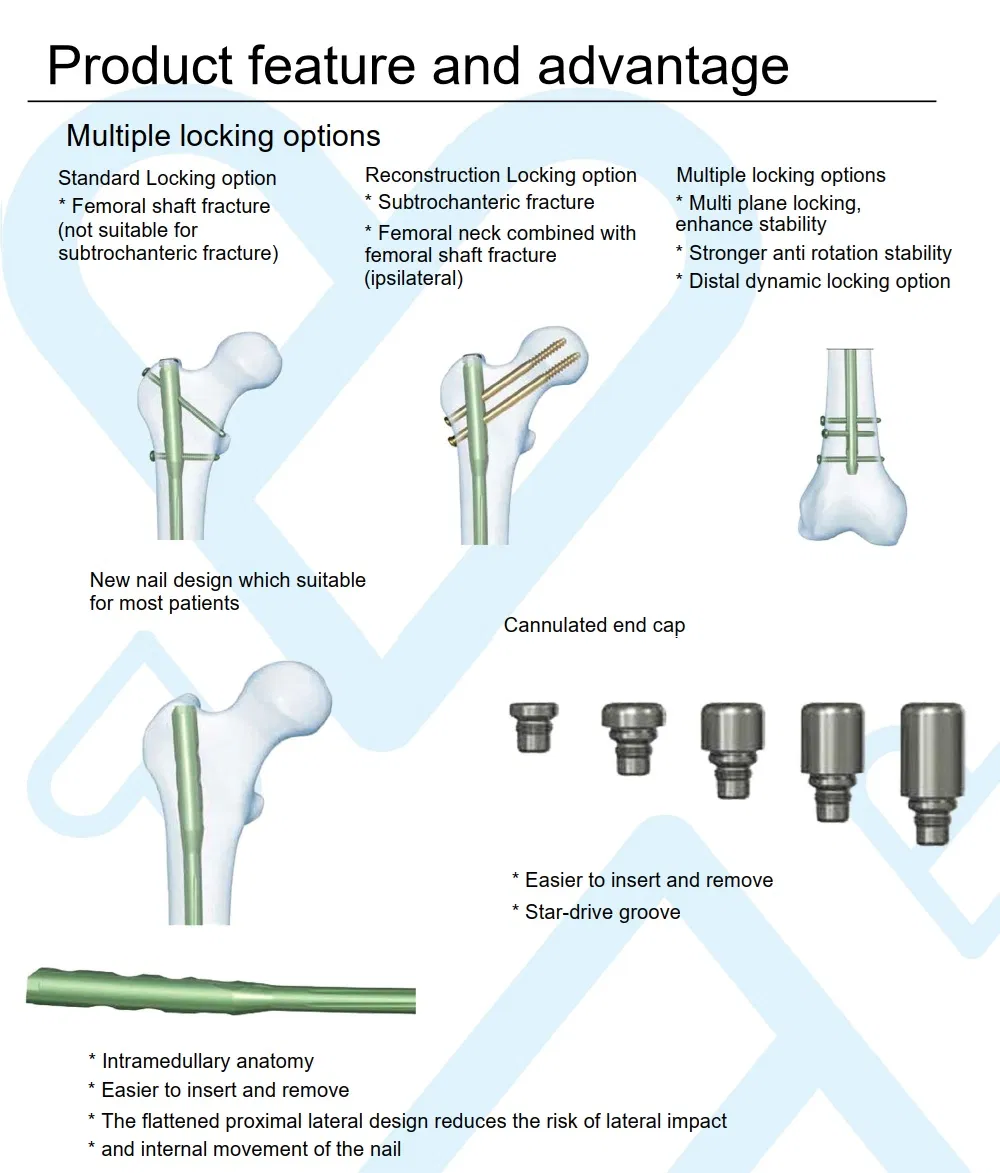
| Expert Femoral Nail | SGG-IIIK | Diameter: 9.0/10.0/11.0mm; Left/Right; Length: 340-400mm | Titanium Alloy (T) |
| Locking Bolt (Large) | SGG-IIIK | Diameter: 6.5mm; Length: 75-100mm | Titanium Alloy (T) |
| Locking Bolt (Small) | SGG-IIIK | Diameter: 5.0mm; Length: 30-80mm | Titanium Alloy (T) |
| End Cap | SGG-IIIK | Length: 0-20mm | Titanium Alloy (T) |
Model: GA3X - Specifically designed for the Expert Femoral Nail System to ensure precision during implantation.









 Nipa Orthopedic Implants
Nipa Orthopedic Implants