

1 / 5










| Still deciding? Get samples of $ ! US$ 80/Piece Request Sample |

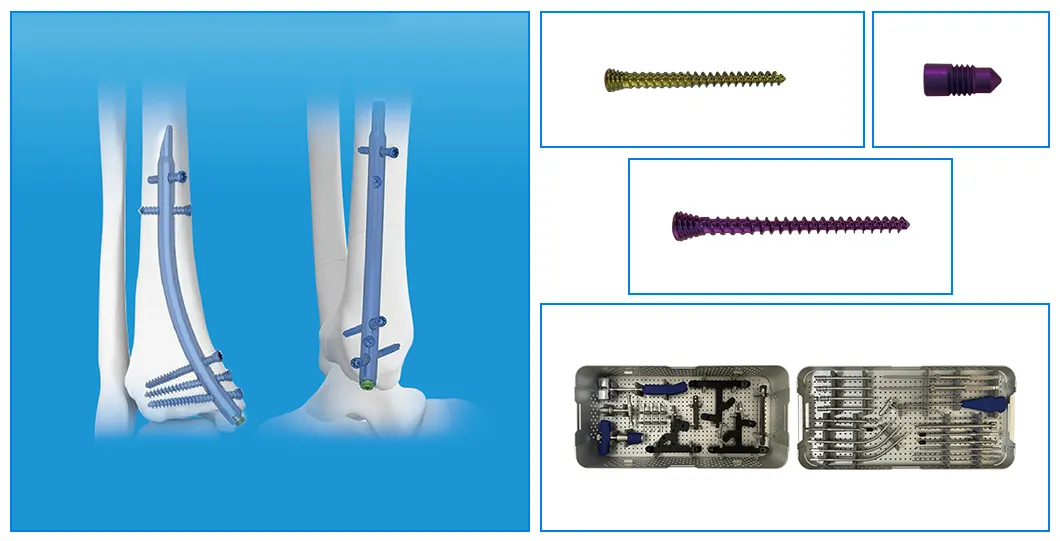
Key Features and Benefits:
| Product Name | Size / Length (mm) | Material |
|---|---|---|
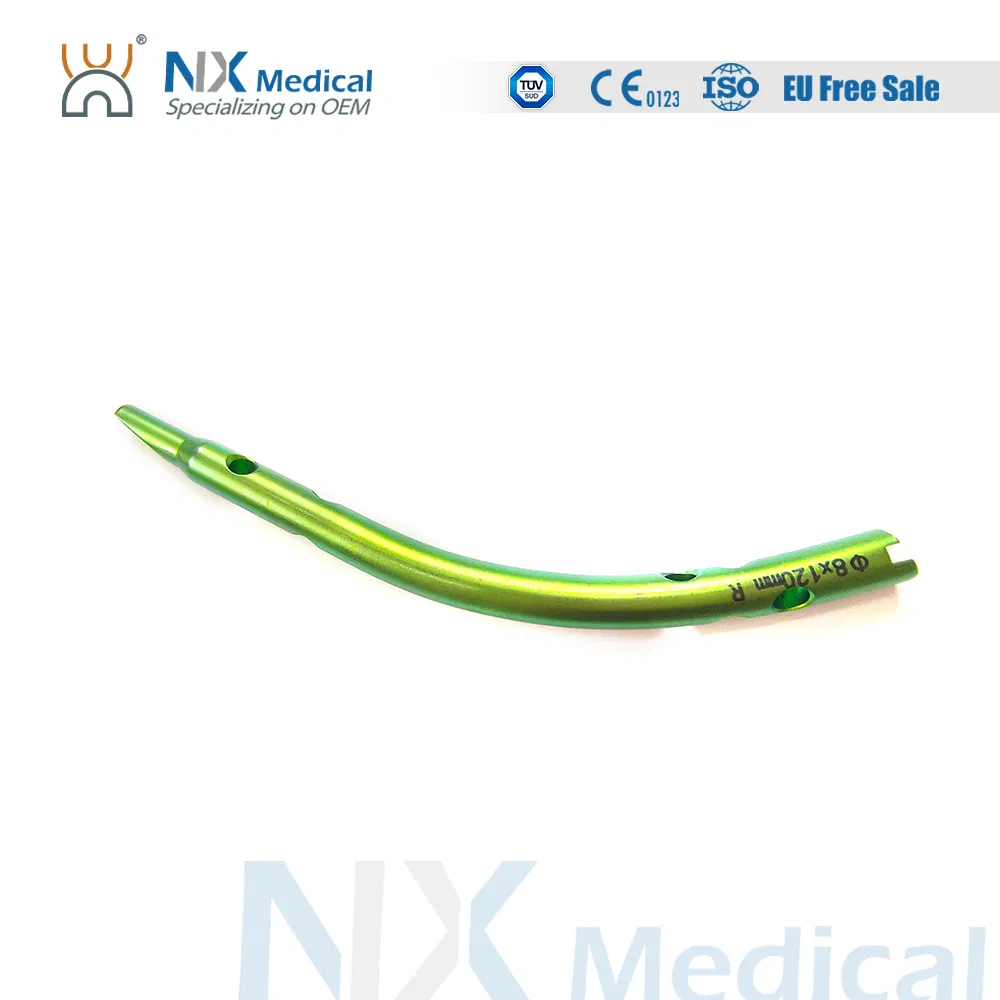
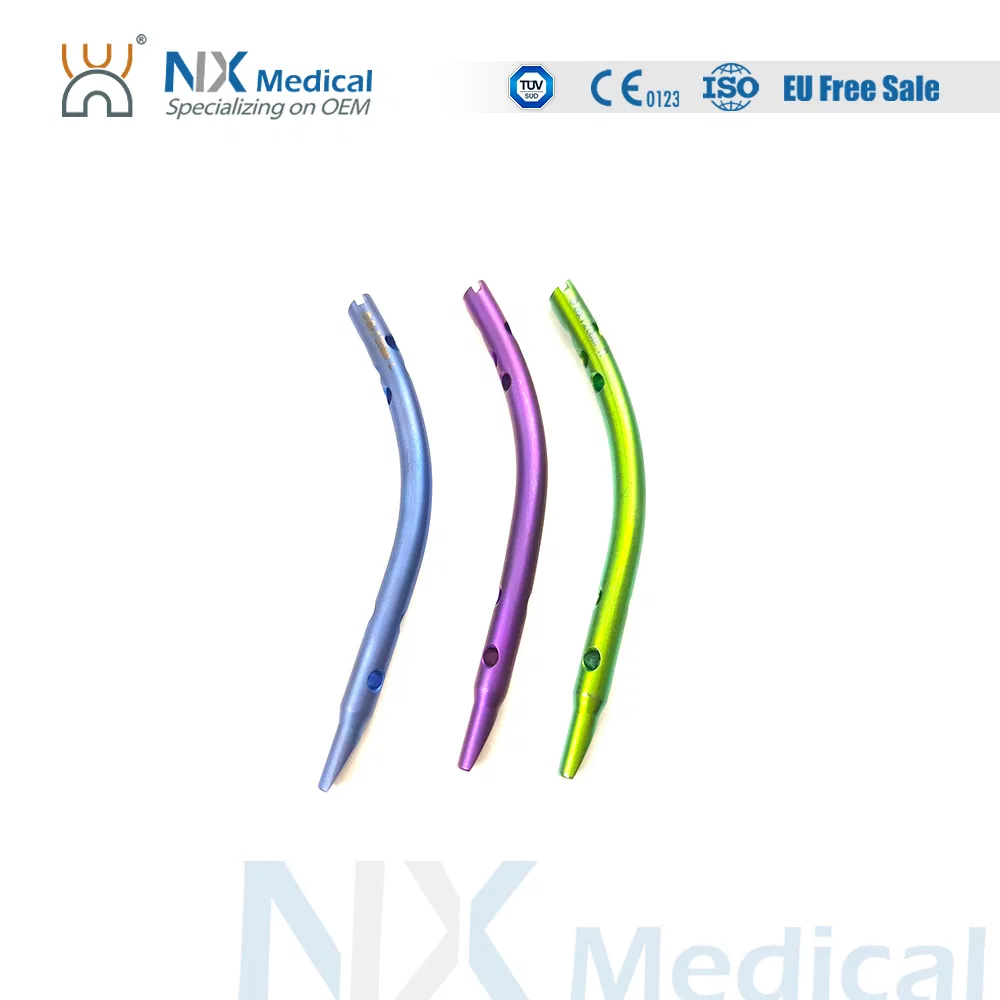
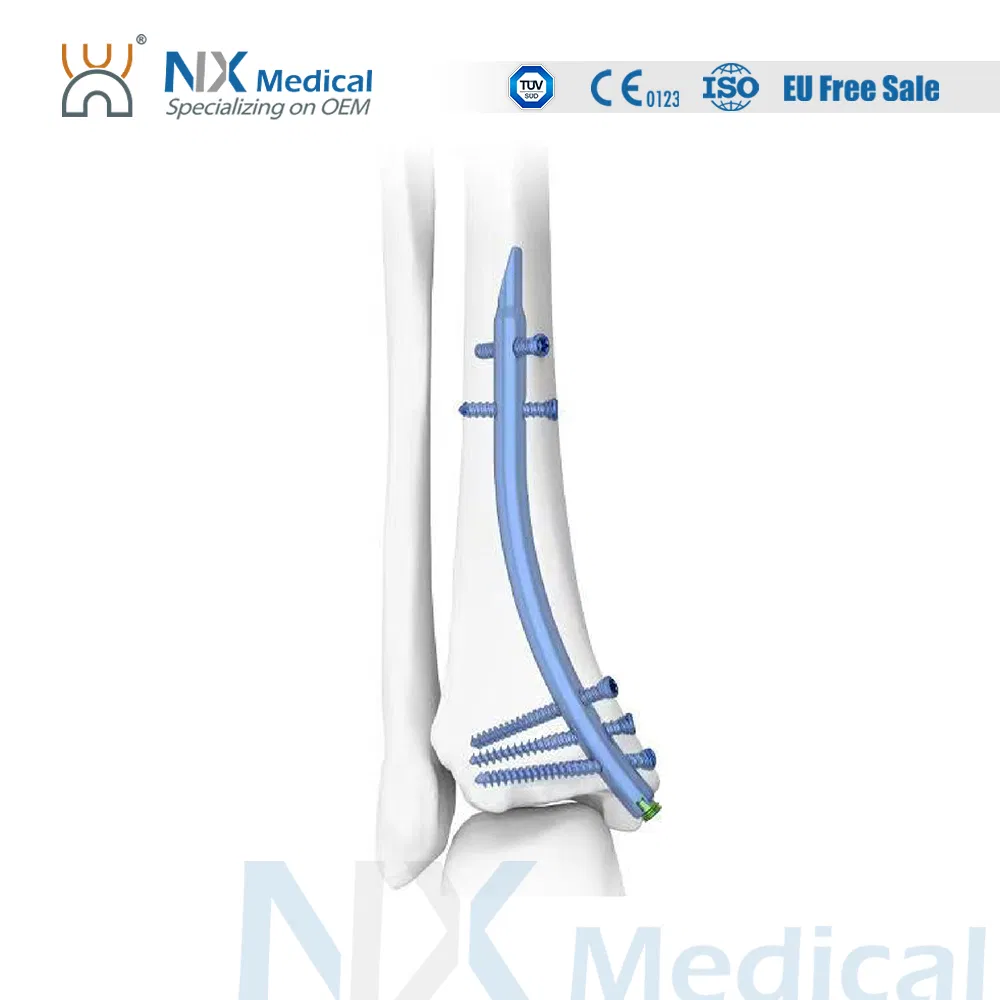
| Retrograde Tibial Nail | 7 x 110 / 8 x 120 / 8 x 140 | Titanium Alloy (TA) |
| φ3.5mm Interlocking Cancellous Screw | 30 / 32 / 34 / 36 / 38 / 40 / 42 / 44 / 46 / 48 / 50 / 52 | Titanium Alloy (TA) |
| φ3.5mm Interlocking Cortical Screw | 18 / 20 / 22 / 24 / 26 / 28 | Titanium Alloy (TA) |
| φ4.0mm Interlocking Cortical Screw | 24 / 26 / 28 / 30 / 32 / 34 | Titanium Alloy (TA) |
| Retrograde Tibial Nail End Cap | Standard / 5mm | Titanium Alloy (TA) |

Our production facilities utilize state-of-the-art CNC machining and rigorous quality control systems. We are committed to providing surgical and reliable therapeutic solutions for orthopedic surgeons and patients worldwide. Our focus remains on high-precision trauma implants and spinal systems certified to international standards.





 Nipa Orthopedic Implants
Nipa Orthopedic Implants